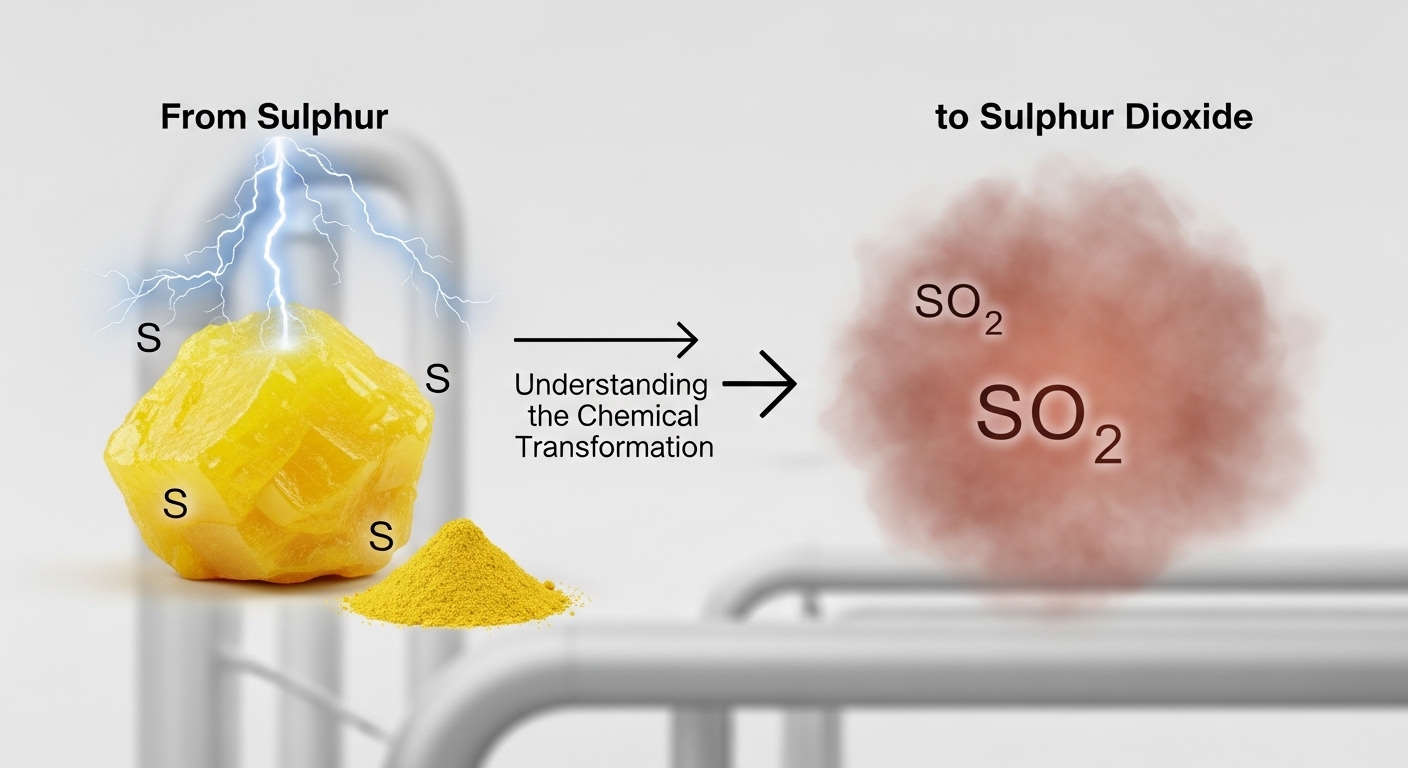
The transformation from sulphur dioxide is a fundamental chemical process that plays a major role in both natural systems and industrial applications. Sulphur is a widely occurring element found in minerals, volcanic emissions, and various chemical compounds. When it reacts with oxygen under suitable conditions, it forms sulphur dioxide, a gas with significant environmental and industrial importance. Understanding how sulphur converts into sulphur dioxide helps explain many chemical reactions that occur in manufacturing, energy production, and atmospheric processes. This transformation illustrates how simple elements interact to form compounds that influence industries, ecosystems, and everyday life around the world.
Understanding the Element Sulphur
Sulphur is a naturally occurring nonmetallic element known for its yellow color and distinctive smell when combined with certain compounds. It exists in many natural forms, particularly in volcanic areas, mineral deposits, and underground resources associated with fossil fuels. The element has been used by humans for centuries in agriculture, medicine, and industrial production. In its pure form, sulphur is relatively stable, but it can easily react with other elements when exposed to heat or oxygen. The reaction from begins when sulphur burns in the presence of oxygen, initiating a chemical transformation that produces a gaseous compound widely studied in chemistry.
The Chemical Reaction from Sulphur to Sulphur Dioxide
The conversion from sulphur to sulphur dioxide occurs through a simple yet important chemical reaction. When sulphur is heated in the presence of oxygen, it undergoes combustion, forming gas. This reaction releases energy in the form of heat and light, making it an exothermic process. Chemically, the reaction involves one sulphur atom combining with two oxygen atoms to create a molecule . The reaction is commonly written in chemical form as sulphur plus oxygen producing . This transformation demonstrates how elemental substances interact with oxygen during combustion to form new chemical compounds with different physical and chemical properties.
Industrial Importance of Sulphur Dioxide Production
The process of converting sulphur to sulphur dioxide has significant industrial value. Sulphur dioxide is an essential intermediate compound used in many manufacturing processes around the world. Industries rely on this gas primarily for the production of sulfuric acid, one of the most important industrial chemicals used in fertilizers, chemical manufacturing, and petroleum refining. The controlled conversion from allows factories to produce large quantities of sulfuric acid efficiently. This process supports agriculture by enabling fertilizer production and supports many sectors that depend on chemical processing, demonstrating how a basic chemical reaction contributes to global industrial development.
Natural Sources of Sulphur Dioxide Formation
The transformation from sulphur to sulphur dioxide also occurs naturally in the environment. One of the most common natural sources is volcanic activity. When volcanoes erupt, they release large amounts of sulphur compounds that react with oxygen in the atmosphere, producing gas. Natural wildfires can also cause the combustion of sulphur-containing materials, contributing to this chemical conversion. Additionally, certain biological processes and geothermal activities release gases that may oxidize. These natural events demonstrate that the reaction from is not limited to industrial settings but also plays an important role in natural atmospheric chemistry.
Environmental Impact of Sulphur Dioxide
Although the conversion from sulphur to sulphur dioxide is useful for industrial production, the resulting gas can also have environmental consequences. Sulphur dioxide released into the atmosphere may react with water vapor to form acidic compounds, contributing to the formation of acid rain. Acid rain can affect soil quality, freshwater ecosystems, forests, and buildings. In high concentrations, may also impact air quality and human health. Because of these concerns, many countries regulate industrial emissions that result from burning sulphur-containing fuels. Understanding the environmental effects of helps governments and industries develop strategies to reduce harmful emissions.
Role in the Production of Sulfuric Acid
One of the most important uses of the reaction from sulphur to sulphur dioxide occurs in the production of sulfuric acid. After forms, it undergoes additional chemical reactions with oxygen and water to create sulfuric acid. This acid is widely used in manufacturing fertilizers, processing metals, producing chemicals, and refining petroleum products. Because sulfuric acid is essential to many industrial operations, the initial conversion from becomes a critical step in a larger chemical process. This connection highlights how a simple reaction can serve as the starting point for producing substances that support global agriculture and industry.
Chemical Properties of Sulphur Dioxide
Is a colorless gas with a strong, sharp odor that is easily recognizable. It dissolves readily in water, forming acidic solutions that influence atmospheric chemistry. The gas acts as a reducing agent in many chemical reactions and can also function as a preservative in certain food processing applications. Understanding the chemical properties of helps scientists and engineers manage its behavior in industrial and environmental systems. The transformation from sulphur to sulphur dioxide therefore introduces a compound with unique characteristics that make it valuable in manufacturing while also requiring careful monitoring to protect environmental health.
Safety and Control in Industrial Processes
Industries that produce sulphur dioxide through the conversion from sulphur to sulphur dioxide must carefully control the process to ensure safety and environmental responsibility. Modern industrial facilities use advanced technologies to capture and process efficiently. Emission control systems help prevent excessive release of the gas into the atmosphere. Workers in these facilities follow strict safety procedures because high concentrations of can be harmful if inhaled. By implementing proper safety measures and pollution control technologies, industries can continue benefiting from the chemical transformation while reducing potential risks to both workers and surrounding communities.
The Scientific Significance of the Reaction
The reaction from is also important in scientific research and education. Chemistry students often study this transformation because it clearly demonstrates the principles of combustion, oxidation, and compound formation. Scientists analyze this reaction to understand how elements behave when exposed to oxygen under high temperatures. The process also provides insight into atmospheric chemistry and environmental science. Because it connects laboratory experiments, industrial applications, and natural phenomena, the transformation from serves as an excellent example of how fundamental chemical reactions influence many aspects of science and everyday life.
Conclusion
The transformation from represents a simple yet powerful chemical process with widespread significance. It occurs naturally in the environment and is also harnessed by industries for essential manufacturing operations. From supporting the production of sulfuric acid to influencing atmospheric chemistry, this reaction demonstrates the interconnected nature of chemical science and real-world applications. While sulphur dioxide plays an important role in industrial development, careful management is necessary to minimize environmental and health impacts. By understanding this transformation, scientists, industries, and policymakers can better balance the benefits of chemical processes with the responsibility of protecting the environment.